COVID-19: Previous AstraZeneca board member defends agency about its dealing with of vaccine rollout | United kingdom News
4 min readA former AstraZeneca board member has advised Sky Information that she thinks the business has been unfairly criticised about its managing of the vaccine rollout.
Professor Dame Nancy Rothwell, who served on the board until finally 5 yrs back, stated she imagined individuals inside of the company would be feeling they have “experienced a bit of a tricky time” as a number of international locations have transformed their guidance on who need to receive the jab.
It will come as the British isles medications regulator claimed this 7 days that the below 30s should be available an different to the AstraZeneca vaccine pursuing stories of pretty rare blood clots.
Stay COVID updates from throughout the United kingdom and all around the environment
Other nations these types of as Germany, Australia and France have given that additional limited who should obtain the AstraZeneca jab.
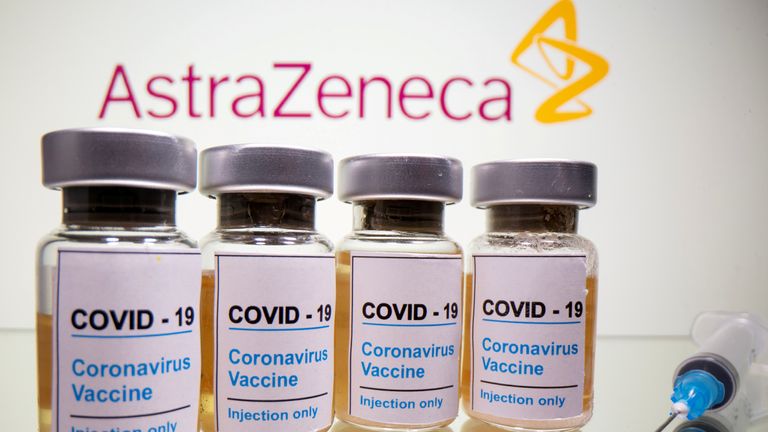
It can be the hottest knock in what has been a bumpy highway for AstraZeneca.
When the pharmaceutical giant struck a offer with Oxford College to develop a COVID-19 vaccine a 12 months ago the enterprise experienced limited vaccine experience. In spite of this it was thought of an early chief in the vaccines race.
It also agreed to manufacture the vaccine at cost not like other providers these kinds of as Pfizer and Moderna which are internet marketing their vaccines at financial gain.
But in the facial area of intense criticism over blunders in its knowledge interaction and offer and supply troubles to the EU, the enterprise has seen a roughly 20% fall in its share selling price considering that Could and a barrage of bad headlines.
Buyers could very well be asking no matter if all the exertion has been value it.
Many close to the company insist it was and that the ambitious move was inspired by a drive to do great.
“It’s been an absolutely enormous achievement,” says Dame Nancy.
“This is a company that is not famous for vaccines and it pretty speedily mobilised. I are not able to imagine how substantially time and exertion it has taken from the executive and the board, all the way as a result of the enterprise to create a vaccine at these very low price tag so speedily, it will have been herculean.
“I suspect some men and women do assume they’ve experienced a bit of a difficult time, I assume I would if I was on the board at the existing time, I would believe blimey, you know we have carried out all this and we’re however receiving some criticism.
“I have spoken to a pair of the executives and they said it is the most complicated issue we have at any time completed, way over and above something we have tried prior to, but I just claimed seem at the tens of millions of lives you’ve got almost certainly saved.”
AstraZeneca has been criticised more than its details conversation. In November it revealed its initial promising trial benefits showing total efficiency prices of 70%, but its publication was followed by confusion and controversy as a few different effectiveness stages experienced been incorporated.
Then in March the results of the vaccine’s US trial ended up disputed by America’s details and basic safety monitoring board, the implication currently being that extra the latest data had been utilized to inflate overall performance.
There has also been a bitter and escalating political row about supply and supply when, in January, AstraZeneca educated the EU it would not be capable to provide all the vaccines it had promised.
Tensions rose as the EU accused the enterprise of prioritising British orders and threatened to block vaccine exports.
“For the up coming several weeks or months, they’re just going to have to remain quiet,” claims Alex Davies who operates Health care PR company Hanover Communications and advises some of the world’s leader pharmaceutical organizations.
“They are heading to have to make absolutely sure that they appropriate misinformation when it appears, but they will need to inform a constant story about the security of their vaccine.
“Reputationally, let’s take a step back again, they have formulated a vaccine, at expense, that will aid the world get back to regular. It truly is likely to get bumpy, but at the close of it, in the very long recreation, they will be remembered as one of the providers that helped save the entire world.”
Nevertheless he and many others worry that AstraZeneca’s experience may have more serious extensive phrase implications.
“If you glimpse at what is actually likely on suitable now, would any other pharma corporation that currently won’t have a vaccine company, adhere to AZ’s route?” he claimed.
“When your share rate is lessen than it was a year in the past and when you happen to be seeing oneself on the front web pages, time and yet again, the answer has to be no which is unfortunate because what AstraZeneca has completed has been brilliant.”
AstraZeneca could hope the next phase will be to roll out to the third world – the small cost and fridge temperature storage make it a fantastic applicant.
But the African Union has stated it is really discovering other solutions with Johnson and Johnson.
The evaluation by the Medicines and Healthcare merchandise Regulatory Agency (MHRA) uncovered that 79 cases of blood clots and 19 fatalities transpired adhering to 20 million doses currently being administered . That presents a chance of about four in a single million of creating a blood clot, and a single in a million of dying.
Each the MHRA and AstraZeneca continue on to strain that the added benefits of the vaccine considerably outweigh the challenges.